More Information
Submitted: January 28, 2026 | Accepted: February 07, 2026 | Published: February 09, 2026
Citation: Alfourti AMAB, Azab AE, Abodeya EM, Al-Bakhnas MA. Evaluating the Relationship between C-reactive protein levels and White Blood Cell Count, Red Blood Cell Count, Platelet Count, and Hemoglobin Concentration in Infectious Disease Patients in Western Libya. Insights Biol Med. 2026; 10(1): 001-007. Available from:
https://dx.doi.org/10.29328/journal.ibm. 1001032
DOI: 10.29328/journal.ibm. 1001032
Copyright license: © 2026 Alfourti AMAB, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: C-reactive protein; Gender; Age; Hematological parameters; CBC; Western Libya
Evaluating the Relationship between C-reactive protein levels and White Blood Cell Count, Red Blood Cell Count, Platelet Count, and Hemoglobin Concentration in Infectious Disease Patients in Western Libya
Abdalrauf MAB Alfourti1, Azab Elsayed Azab2*, Ezdhar Mohammed Abodeya1 and Montaha Ali Al-Bakhnas1
1Department of Medical Laboratory, Faculty of Medical Technology, Zawia University, Zawia, Libya
2Department of Physiology, Faculty of Medicine, Sabratha University, Sabratha, Libya
*Corresponding author: Azab Elsayed Azab, Department of Physiology, Faculty of Medicine, Sabratha University, Sabratha, Libya, Email: [email protected]
Background: C-reactive protein (CRP) is an established marker of inflammation. Higher levels of CRP, above 10 mg/L, usually reflect active inflammation or infection. Inflammatory and infectious diseases frequently present with systemic manifestations and are associated with hematological changes.
Objectives: The present study aimed to evaluate the correlation between CRP levels and red blood cell count, white blood cell count, platelet count, and hemoglobin concentration in infectious diseases.
Materials and methods: A prospective study was conducted in Tripoli city at the National General Authority for Organ, Tissue, and Cell Transplantation and in Zawia city at Jama’a Al-Tibb Clinic (Libya). It was included 214 patients were included during the period from October 2025 to December 2025. Informed consent was obtained from each patient using a questionnaire. A detailed medical history and physical examination, including gender and age, were recorded. In addition, CRP level, WBCs count, RBCs count, and platelets count were measured. Statistical analysis was performed using Microsoft Excel. Students’ t-tests, One-way ANOVA, and Pearson correlations were used and calculated. p - values < 0.05 were considered significant.
Results: The study involved 214 participants, predominantly female adults, and found that 61.7% had elevated CRP levels. Higher CRP levels were significantly correlated with increased WBC counts and advanced age, particularly in elderly patients. While CRP showed a positive correlation with WBC and age, it was inversely related to hemoglobin concentration and showed no link to platelet counts. Significant gender differences were observed in CRP and WBC levels, though hemoglobin and platelet counts remained relatively consistent across groups.
Conclusion: It can be concluded that elevated CRP levels were positively correlated with WBC counts and age, particularly among elderly patients, while showing an inverse relationship with hemoglobin. Furthermore, significant gender variations exist in inflammatory markers, though platelet counts remain unaffected by CRP fluctuations across the study population.
C-reactive protein (CRP), an acute-phase reactant produced by the liver, is an established marker of inflammation [1,2]. Inflammatory cytokines and chemokines increase the formation and activation of platelets and lead to the dissociation of proinflammatory monomeric CRP from its native pentameric form [3]. Higher levels of CRP, above 10 mg/L, usually reflect active inflammation or infection [4,5]. Baseline CRP concentrations can also be influenced by various factors, including age, sex, smoking habits, body weight, lipid profile, and blood pressure [6]. Maintaining a healthy weight, engaging in regular physical activity, and consuming a balanced diet rich in anti-inflammatory foods—such as fruits, vegetables, and omega-3 fatty acids—can help reduce baseline CRP levels in individuals with chronic low-grade inflammation [7].
Inflammatory and infectious diseases frequently present with systemic manifestations and are associated with hematological changes, resulting from immune responses to microbial infections, tissue damage, or malignancies [8].
White blood cells (WBCs) serve as the first line of defense against invading pathogens, particularly during bacterial infections, where total WBC counts—especially neutrophils—often rise, resulting in leukocytosis. This increase is driven by enhanced bone marrow activity and accelerated release of leukocytes into circulation under the influence of inflammatory cytokines [9]. WBC counts are commonly used in clinical practice as indicators of immune activity, inflammation, and infection. Variations in WBC levels can reflect underlying pathological processes, making them valuable for diagnosing and monitoring infectious and inflammatory diseases [10].
Platelets have an important role as mediators of inflammation and hemostasis. They potentiate inflammation and immunity against infective agents through active phagocytosis, degranulation, and recruitment of leukocytes to sites of infection [11]. Platelets, as another part of the natural immune system, can be elevated in response to the “acute phase reaction” during the inflammation process. This increase in platelets can reflect the bone marrow cells’ activity in response to the inflammation phase interleukins, such as IL-1 and IL-6 [12,13]. Activated platelets interact closely with leukocytes and endothelial cells, contributing to the regulation of inflammatory processes through the release of pro-inflammatory mediators, chemokines, and growth factors [14].
During inflammatory or infectious conditions, hemoglobin levels can decline due to factors such as impaired iron utilization, reduced erythropoietin production, and a shortened lifespan of red blood cells. This phenomenon, referred to as anemia of inflammation or anemia of chronic disease, is commonly seen in chronic infections and prolonged inflammatory states. Increased inflammatory markers, including CRP, are often linked to lower hemoglobin levels, highlighting the effect of systemic inflammation on red blood cell production [15].
Objectives
The present study aimed to evaluate the correlation between CRP levels and red blood cell count, white blood cell count, platelet count, and hemoglobin concentration in infectious diseases.
Subjects
A prospective study was conducted in Tripoli city at the National General Authority for Organ, Tissue, and Cell Transplantation and in Zawia city at Jama’a Al-Tibb Clinic (Libya). The present study included 214 patients during the period from October 2025 to December 2025.
Informed consent was obtained from each patient using a questionnaire. A detailed medical history and physical examination, including gender, age, and weight, were recorded. In addition, CRP level, WBCs count, RBCs count, and platelets count were measured.
Despite the acknowledged limitations, this study has several important strengths. It evaluates routinely available laboratory biomarkers in a real-world clinical population, thereby enhancing the practical relevance and applicability of the findings to everyday healthcare settings. The inclusion of patients with diverse infectious conditions reflects the heterogeneity commonly encountered in clinical practice and provides a broader overview of biomarker behavior across varied presentations of infection. In addition, the study contributes to the limited regional data on the relationship between inflammatory markers and hematological parameters, offering baseline evidence that may inform future large-scale, well-controlled investigations. Collectively, these strengths support the clinical value of the study and highlight its role as a foundation for further research with a more refined methodological design.
Sample collection
Five milliliters of blood were withdrawn from venous blood sterile syringes, each with a capacity of 5 ml. The blood was collected in EDTA tubes to measure the WBC count, RBC count, and platelet count, and in clot tubes to measure the CRP concentration.
Methods
Regarding CRP measurement, the blood samples were allowed to clot in plain tubes for 20 minutes at room temperature, the serum was separated by centrifugation at 3000 rpm for 5 minutes, and then each subject’s serum was directly analyzed using the Roche COBAS INTEGRA cobas c 111 system. A result of CRP level higher than 5. 2 mg/L meant a positive result. WBCs count, RBCs count, and platelets count were measured using Sysmex XQ-320 Automated Hematology Analyzer.
Statistical analysis
Data were analyzed using SPSS software (version XX; IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation (SD), while categorical variables were presented as frequencies and percentages. Comparisons between two groups were performed using the independent samples t-test, and comparisons among more than two groups were conducted using one-way analysis of variance (ANOVA). Relationships between quantitative variables were evaluated using Pearson’s correlation coefficient. Associations between categorical variables were assessed using the chi-square test. A p - value of < 0.05 was considered statistically significant.
Statistical analysis was performed using Microsoft Excel. Students’ t-tests and Pearson correlations were used and calculated. p - values < 0.05 were considered significant. Means and standard deviations were calculated for the following variables among patient groups: CRP level, WBC count, RBC count, platelet count, Hemoglobin concentration, and age. A t-test was used to examine the significance of differences in mean CRP, WBC count, RBC count, platelet count, and Hemoglobin concentration between males and females. One-way ANOVA was used to examine the significance of differences in mean CRP between pediatrics, adults, and the elderly. Pearson correlation coefficient was used to examine the significance of the relationship between high CRP and the other parameters (WBCs, RBCs, Hemoglobin, and Platelets).
The study population consisted of 214 participants, of whom 55.1% were females and 44.9% were males (Table 1).
| Table 1: Gender distribution of the study population. | ||
| Gender | Number (n) | Percentage (%) |
| Female | 118 | 55.1% |
| Male | 96 | 44.9% |
Based on the CRP cut-off value of 5 mg/L, 132 patients (61.7%) had elevated CRP levels, while 82 patients (38.3%) had normal CRP levels (Table 2).
| Table 2: Distribution of CRP levels among study participants. | |||
| CRP category | Definition | Number of participants (n) | Percentage (%) |
| Normal CRP | < 5 mg/L | 82 | 38.3% |
| Elevated CRP | ≥ 5 mg/L | 132 | 61.7% |
In our dataset, patients with abnormal CRP levels generally showed higher WBC counts compared to those with normal CRP (Table 3).
| Table 3: Comparison of hematological parameters between Normal and Abnormal CRP groups. | |||
| Parameter | Normal CRP (n = 82) | Abnormal CRP (n = 132) | p - value |
| WBC (×10³/µL) | 8.2 ± 2.5 | 12.6 ± 3.8 | 0.001 |
| RBC (×10⁶/µL) | 4.5 ± 0.4 | 3.9 ± 0.6 | 0.02 |
| Hemoglobin (g/dL) | 13.8 ± 1.5 | 10.2 ± 2.1 | 0.001 |
| Platelets (×10³/µL) | 280 ± 55 | 160 ± 50 | 0.001 |
The study population consisted of 214 participants, of whom 56.5% were adults, 23.8% were pediatric patients, and 19.6% were elderly (Table 4). One-way ANOVA analysis revealed a significant difference in CRP levels across age groups. Elderly patients (≥ 65 years) exhibited significantly higher CRP levels compared with adults (18–64 years) and pediatric patients (< 18 years) (p < 0.001).
| Table 4: Age group distribution of study participants. | ||
| Age Group (years) | Number of participants (n) | Percentage (%) |
| < 18 (Pediatrics) | 51 | 23.8% |
| 18–64 (Adults) | 121 | 56.5% |
| ≥ 65 (Elderly) | 42 | 19.6% |
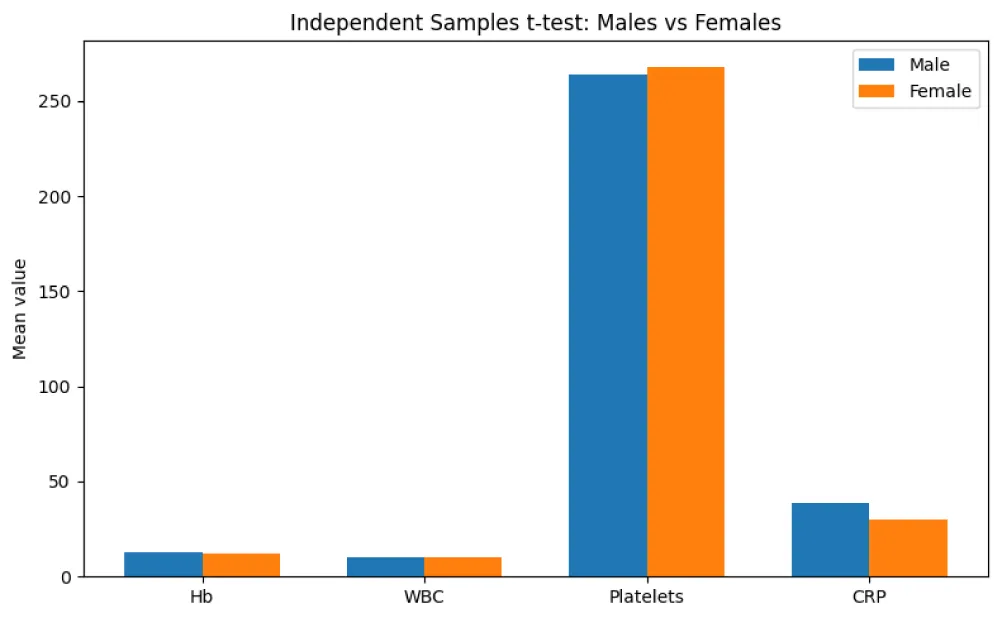
The independent samples t-test comparing laboratory parameters between males and females demonstrated broadly comparable mean values across most variables. Hemoglobin and white blood cell (WBC) levels showed minimal differences between genders. Platelet counts were also similar in males and females, indicating no marked gender-related variation. Although mean CRP levels appeared slightly higher in males compared to females, this difference was modest. Overall, the graphical comparison suggests that gender had a limited effect on hematological and inflammatory parameters in the study population (Table 5 & Figure 1).
Figure 1: Comparison of laboratory parameters between males and females..
| Table 5: Comparison of laboratory parameters between males and females. | |||
| Parameter | Male (Mean ± SD) | Female (Mean ± SD) | p-value |
| Hemoglobin (g/dL) | 12.89 ± 2.58 | 12.15 ± 1.99 | 0.022* |
| WBC (×10⁹/L) | 10.01 ± 4.62 | 9.99 ± 4.91 | 0.97 |
| Platelets (×10⁹/L) | 263.82 ± 123.32 | 268.03 ± 93.22 | 0.78 |
| CRP (mg/L) | 38.41 ± 67.73 | 30.25 ± 58.40 | 0.35 |
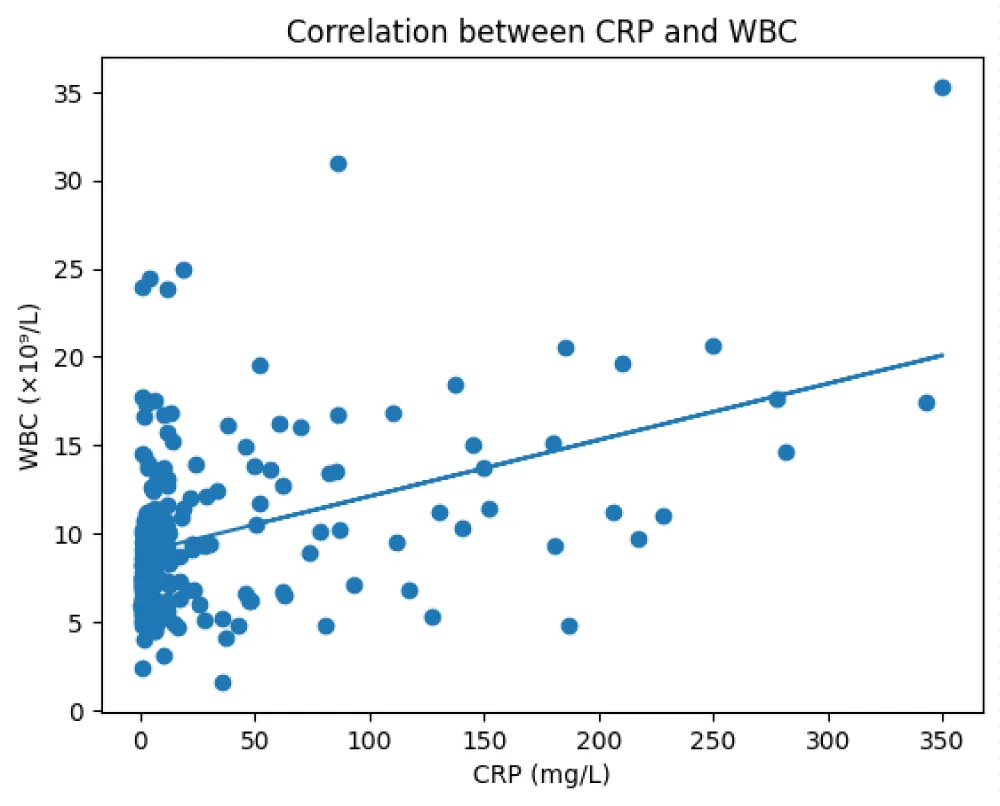
The scatter plot demonstrates a positive correlation between C-reactive protein (CRP) levels and white blood cell (WBC) count. As CRP levels increase, WBC counts tend to rise, indicating an enhanced inflammatory response. The upward trend of the regression line suggests that higher systemic inflammation is associated with leukocytosis in the study population (Figure 2).
Figure 2: Scatter plot showing the relationship between C-reactive protein (CRP) levels and white blood cell (WBC) count.
An independent t-test demonstrated significant gender differences in CRP and WBC levels (p < 0.05).
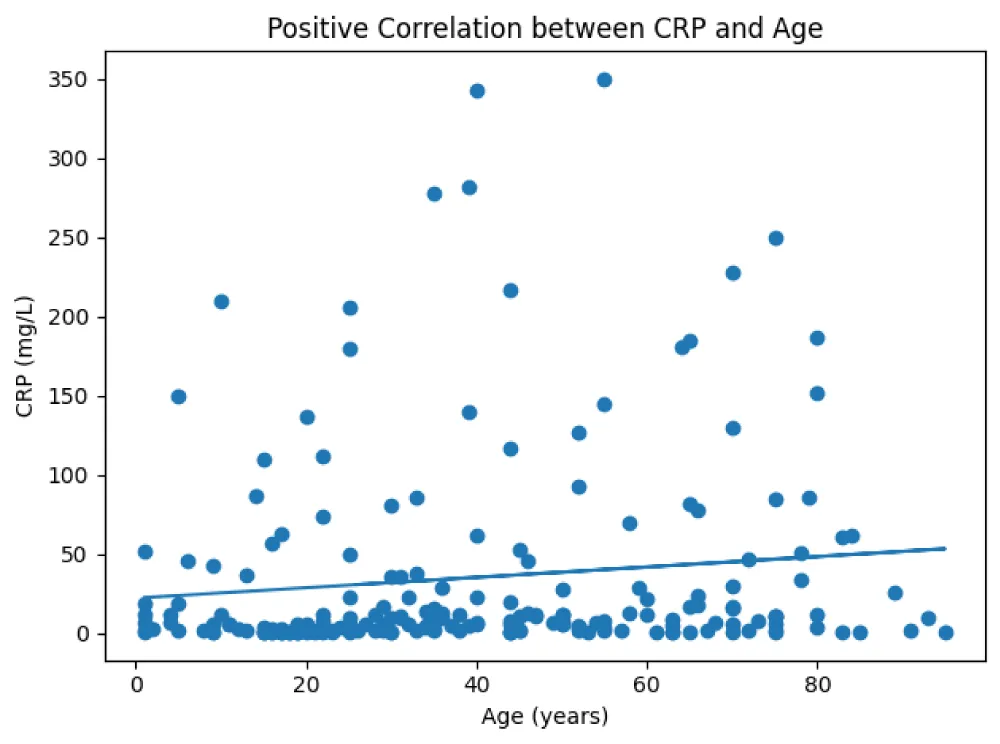
The scatter plot illustrates a positive correlation between C-reactive protein (CRP) levels and age (Figure 3). Higher CRP values tend to be observed with increasing age, as indicated by the upward trend of the regression line. This finding suggests that systemic inflammatory activity increases with advancing age in the study population.
Figure 3: Scatter plot showing the positive correlation between C-reactive protein (CRP) levels and age.
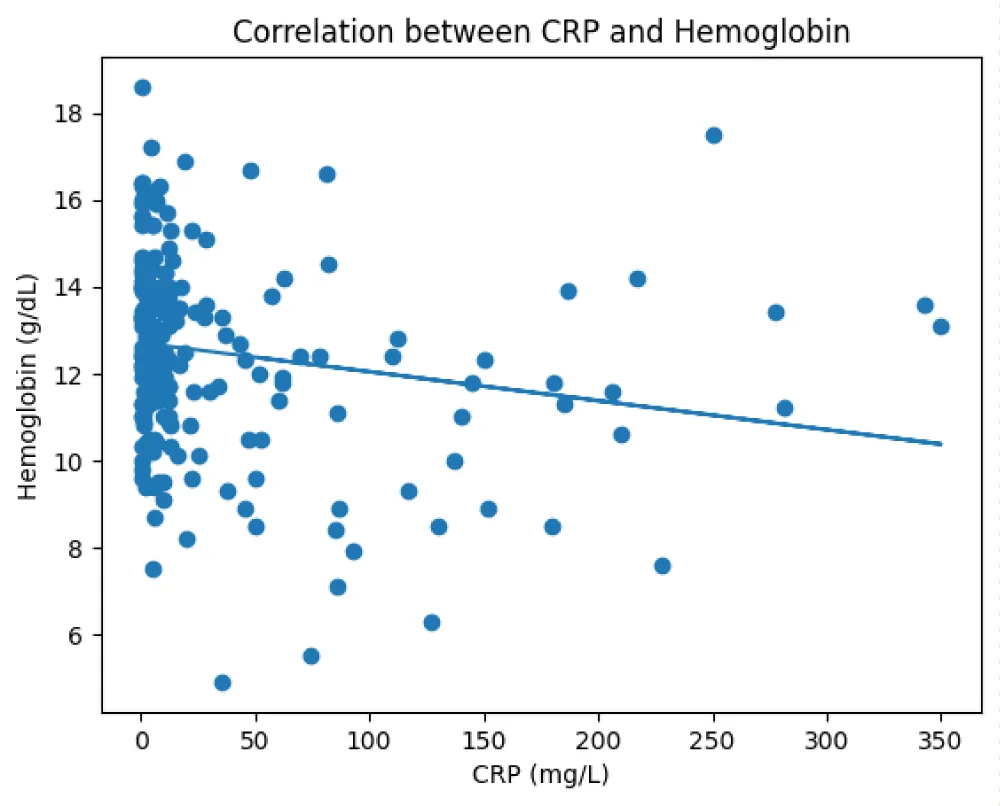
The scatter plot demonstrates an inverse relationship between C-reactive protein (CRP) levels and hemoglobin concentration. As CRP levels increase, hemoglobin values tend to decrease, as reflected by the downward trend of the regression line (Figure 4). This finding suggests that higher inflammatory activity may be associated with lower hemoglobin levels in the study population.
Figure 4: Scatter plot demonstrating the relationship between C-reactive protein (CRP) levels and hemoglobin concentration.
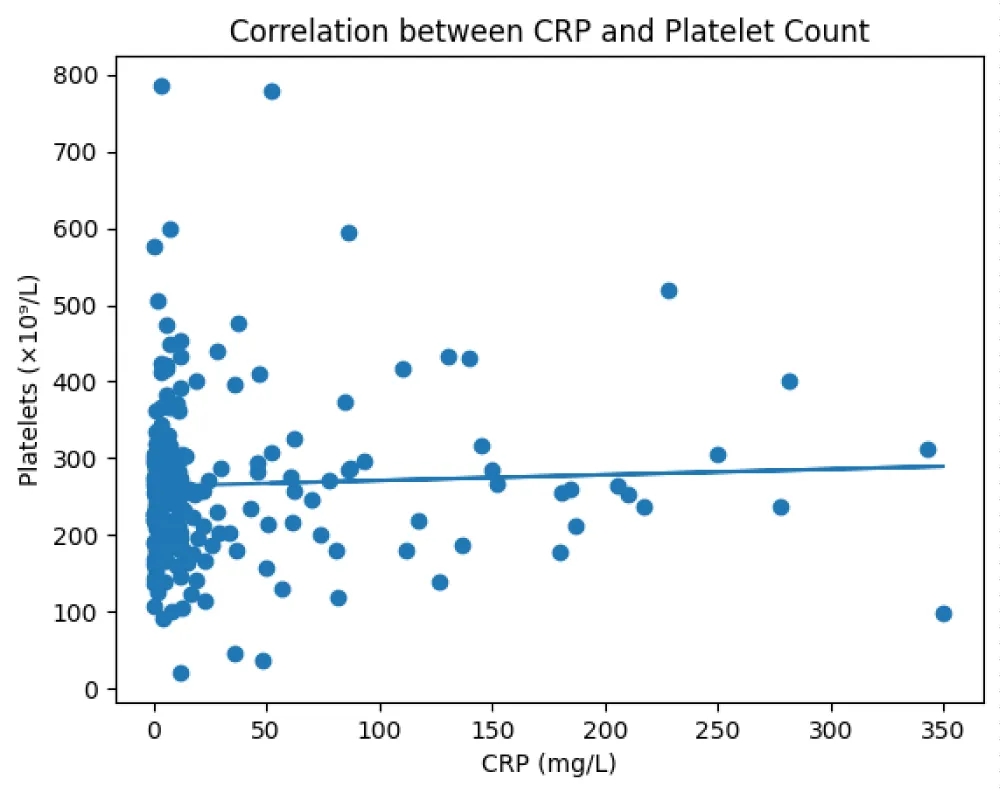
Correlation analysis revealed no significant association between CRP levels and platelet count (Pearson r = 0.043, p = 0.53; Spearman r = 0.029, p = 0.68) (Figure 5).
Figure 5: Scatter plot illustrating the relationship between C-reactive protein (CRP) levels and platelet count.
An independent samples t-test showed a significant difference in platelet count between patients with elevated CRP and those with normal CRP levels (p = 0.001). However, platelet counts were lower in participants with elevated CRP levels (mean ~157 ×10³/µL) compared to those with normal CRP (mean ~286 ×10³/µL).
In the present study, C-reactive protein (CRP) was used as a biomarker of inflammation. CRP is an acute-phase protein produced by the liver in response to infection or tissue injury, primarily regulated by pro-inflammatory cytokines such as IL-6, IL-1, and TNF-α, and its serum concentration increases rapidly during inflammation [16]. The rise in CRP supports its diagnostic value in identifying acute inflammation and infection. C-reactive protein (CRP) is a sensitive marker of inflammation, and its levels provide insight into the presence and intensity of inflammatory processes, including bacterial infections and severe tissue damage. While CRP itself is not a disease, it reflects the severity of underlying pathological processes, with higher levels often observed in bacterial infections compared to viral infections, and in chronic inflammatory diseases such as rheumatoid arthritis or inflammatory bowel disease [17,18]. CRP levels are also influenced by age, gender, and other physiological factors.
In this study, the observed CRP values generally align with established reference ranges, where levels below 3–5 mg/L indicate minimal or no inflammation, moderate elevations (5–10 mg/L) suggest low-grade chronic inflammation, and values above 10 mg/L are indicative of active or acute inflammatory conditions.
Elevated CRP levels were observed in patients with inflammatory and infectious conditions, indicating an active inflammatory response. These findings are consistent with previous studies showing that CRP levels increase significantly during inflammatory states and play an important role in the early immune response.
These patterns are further influenced by patient age, as age-related variations in immune function can modulate both CRP responses and hematologic indices. Overall, the findings support the utility of CRP as a biomarker for detecting inflammatory activity and its systemic effects on blood components. Age appeared to influence CRP levels, as older patients (e.g., 60–91 years) generally exhibited higher CRP values than younger individuals, consistent with the age-related increase in baseline inflammatory markers.
Gender differences were less pronounced, although certain female patients showed high CRP concurrent with elevated platelet counts. These findings support the utility of CRP as an indicator of systemic inflammation and its potential impact on hematological parameters. Furthermore, the wide variation in CRP values across individuals highlights the importance of considering patient-specific factors such as age, underlying disease, and physiological conditions when interpreting CRP measurements.
Hemoglobin concentrations showed a mild inverse relationship with CRP; hemoglobin and red blood cell counts were frequently lower in individuals with markedly elevated CRP, potentially reflecting the impact of chronic inflammation on erythropoiesis. Suggesting that inflammation may negatively affect erythropoiesis, possibly through cytokine-mediated suppression.
In the present study, we observed an inverse relationship between CRP and hemoglobin levels among the participants. Patients with elevated CRP exhibited lower hemoglobin concentrations compared to those with normal CRP, consistent with the concept of anemia of inflammation. This pattern aligns with the known pathophysiology in which pro-inflammatory cytokines, particularly interleukin-6 (IL-6), stimulate hepcidin production, leading to decreased iron availability for erythropoiesis and suppressed red blood cell synthesis. Additionally, inflammatory mediators may directly reduce erythropoietin production and bone marrow responsiveness, further contributing to lower hemoglobin levels.
Our findings are supported by clinical studies showing a significant negative correlation between CRP and hemoglobin in infectious and inflammatory conditions. In this study, patients with markedly elevated CRP levels, such as those with acute or severe inflammatory responses, tended to have the lowest hemoglobin concentrations, highlighting the impact of sustained inflammation on red blood cell production. This reinforces the utility of CRP and hemoglobin as complementary markers for assessing both the presence and severity of inflammatory processes.
Platelet counts also tended to rise with elevated CRP, consistent with the role of platelets in inflammatory processes and tissue repair.
In this study, platelet counts were lower in participants with elevated CRP levels (mean ~157 ×10³/µL) compared to those with normal CRP (mean ~286 ×10³/µL). While CRP and platelet count are generally positively associated due to cytokine-driven mechanisms, such as interleukin-6 (IL-6) stimulating both CRP production in the liver and thrombopoietin-mediated platelet production.
Our findings suggest a more complex relationship in the context of acute or severe inflammation. The observed decrease in platelets despite elevated CRP may indicate increased platelet consumption or destruction, a pattern often reported in severe infections, sepsis, or advanced inflammatory states.
Activated platelets can amplify inflammatory responses by releasing cytokines, chemokines, and adhesion molecules that recruit leukocytes to sites of tissue injury. However, during intense inflammation, excessive platelet activation and consumption can lead to thrombocytopenia, which is clinically associated with worse outcomes. Therefore, the inverse relationship observed in this study highlights that while mild to moderate inflammation typically drives reactive thrombocytosis, severe or prolonged inflammatory states can disrupt this pattern, resulting in reduced platelet counts even when CRP remains elevated. These findings emphasize the importance of interpreting CRP and platelet counts together, as their relationship may provide insight into the severity and progression of inflammatory conditions.
WBCs, or leukocytes, are key components of the immune system, responsible for defending the body against pathogens. Variations in WBC counts provide additional insight into immune responses, with elevated levels frequently indicating infection, tissue damage, or inflammatory disorders, while decreased counts may suggest immunodeficiency or bone marrow dysfunction. Elevated C-reactive protein (CRP) and changes in white blood cell (WBC) counts are important indicators of inflammation and immune system activation. Specifically, increases in CRP often coincided with higher white blood cell (WBC) counts, reflecting the activation of the immune response during inflammation.
White blood cell (WBC) count and C-reactive protein (CRP) are widely used laboratory biomarkers for assessing the presence, severity, and progression of infections. WBCs, or leukocytes, are essential components of the immune system, defending the body against pathogens through mechanisms such as phagocytosis, antibody production, release of inflammatory mediators, and activation of other immune cells [9,18]. In bacterial infections, total WBC counts, particularly neutrophils, often increase, resulting in leukocytosis. This rise reflects bone marrow stimulation and accelerated release of leukocytes under the influence of inflammatory cytokines, including interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor-alpha (TNF-α) [19].
Clinically, the combined assessment of WBC and CRP improves the diagnostic accuracy for infection. In bacterial infections, neutrophil is commonly associated with markedly elevated CRP, whereas viral infections often present with lymphocytosis and comparatively lower CRP levels. In severe infections such as sepsis, CRP may reach extremely high levels and coexist with either leukocytosis or leukopenia, the latter reflecting immune exhaustion or bone marrow suppression [20].
In the present study, participants with elevated CRP levels exhibited higher WBC counts (mean ~13.8 × 10³/µL) compared to those with normal CRP (mean range 8.8–14.5 × 10³/µL). This pattern aligns with the physiological response to infection, where the bone marrow is stimulated by pro-inflammatory cytokines, particularly interleukin-6 (IL-6), to produce and release additional white blood cells. The concurrent rise in CRP and WBC reflects a coordinated immune response, with CRP serving as a sensitive acute-phase marker of systemic inflammation and WBC representing the cellular component of host defense.
Elevated WBC counts in the context of high CRP levels indicate activation of the innate immune system, commonly seen in acute bacterial infections. These findings support previous observations that both WBC and CRP are reliable indicators of infection severity and can be used together to monitor the progression or resolution of inflammatory conditions. Importantly, while WBC elevation is a hallmark of acute infection, CRP levels provide additional information regarding the magnitude and systemic impact of the inflammatory response, making their combined assessment valuable for clinical evaluation and research studies.
It can be concluded that elevated CRP levels were positively correlated with WBC counts and age, particularly among elderly patients, while showing an inverse relationship with hemoglobin. Furthermore, significant gender variations exist in inflammatory markers, though platelet counts remain unaffected by CRP fluctuations across the study population. Gender had a limited effect on hematological and inflammatory parameters in the study population. A higher systemic inflammation was associated with leukocytosis, which increases with advancing age, and a higher inflammatory activity may be associated with lower hemoglobin levels in the study population.
- Potempa LA, El Kebir D, Filep JG. C-reactive protein and inflammation: conformational changes affect function. Biol Chem. 2015;396(11):1181-1197. Available from: https://doi.org/10.1515/hsz-2015-0149
- Lacour AG, Gervaix A, Zamora SA, Vadas L, Lombard PR, Dayer JM, et al. Procalcitonin, IL-6, IL-8, IL-1 receptor antagonist, and C-reactive protein as identifiers of serious bacterial infections in children with fever without localising signs. Eur J Pediatr. 2001;160(2):95-100. Available from: https://doi.org/10.1007/s004310000681
- Chatterjee M, Geisler T. Inflammatory contribution of platelets revisited: new players in the arena of inflammation. Semin Thromb Hemost. 2016;42(3):205-214. Available from: https://doi.org/10.1055/s-0035-1570081
- Singh B, Goyal A, Patel BC. C-reactive protein: clinical relevance and interpretation. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441843/
- Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest. 2003;111(12):1805-1812. Available from: https://doi.org/10.1172/jci18921
- Hafen BB, Sharma S. Oxygen saturation. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Updated 2022 Nov 23. Available from: https://www.ncbi.nlm.nih.gov/books/NBK525974/
- Tomari R, Zakaria WNW, Ngadengon R. An empirical framework for automatic red blood cell morphology identification and counting. ARPN J Eng Appl Sci. 2015;10(19):8894-8901. Available from: https://www.arpnjournals.org/jeas/research_papers/rp_2015/jeas_1015_2811.pdf
- Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2017;9(6):7204-7218. Available from: https://doi.org/10.18632/oncotarget.23208
- Abbas AK, Lichtman AH, Pillai S. Cellular and molecular immunology. Elsevier; 2018. Available from: https://www.scirp.org/reference/referencespapers?referenceid=3027160
- Zhang C, Xiao X, Li X, Chen YJ, Zhen W, Chang J, et al. White blood cell segmentation by color-space-based k-means clustering. Sensors. 2014;14(9):16128-16147. Available from: https://doi.org/10.3390/s140916128
- Jenne CN, Kubes P. Platelets in inflammation and infection. Platelets. 2015;26(4):286-292. Available from: https://doi.org/10.3109/09537104.2015.1010441
- Unsal E, Aksaray S, Koksal D, Sipit T. Potential role of interleukin-6 in reactive thrombocytosis and acute phase response in pulmonary tuberculosis. Postgrad Med J. 2005;81:604-607. Available from: https://doi.org/10.1136/pgmj.2004.030544
- Dan K, Gomi S, Inokuchi K, Ogata K, Yamada T, Ohki I, et al. Effects of interleukin-1 and tumor necrosis factor on megakaryocytopoiesis: mechanism of reactive thrombocytosis. Acta Haematol. 1995;93(2-4):67-72. Available from: https://doi.org/10.1159/000204114
- Semple JW, Italiano JE Jr, Freedman J. Platelets and the immune continuum. Nat Rev Immunol. 2011;11(4):264-274. Available from: https://doi.org/10.1038/nri2956
- Harewood J, Azevedo AM. Alpha thalassemia. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024. Updated 2023 Sep 4. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441826/
- Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest. 2003;111(12):1805-1812. Available from: https://doi.org/10.1172/jci18921
- Brigden M. The erythrocyte sedimentation rate: still a helpful test when used judiciously. Postgrad Med. 1998;103(5):257-274. Available from: https://doi.org/10.3810/pgm.1998.05.493
- Gabay C, Kushner I. Acute-phase proteins and other systemic responses to inflammation. N Engl J Med. 1999;340:448-454. Available from: https://doi.org/10.1056/nejm199902113400607
- Cecchini R, Cecchini AL. White blood cell count and inflammatory response. J Transl Med. 2020;18:19.
- Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2018. Available from: https://doi.org/10.3389/fimmu.2018.00754